Understanding the Molecular Pathways of
GI Cancer: Insights into Diagnosis and Targeted Therapies
Gastrointestinal (GI) cancers encompass a heterogeneous group of malignancies, including colorectal, gastric, pancreatic, esophageal, and hepatobiliary cancers. Advances in molecular oncology have significantly enhanced our understanding of the pathways driving tumorigenesis, offering new avenues for precision diagnosis and targeted therapies.
Key Molecular Pathways in GI Cancers
RAS/RAF/MEK/ERK Pathway, Claudin 18.2, HER2 Amplification, EGFR/ FGFR2, c-MET, TKI/ VEGF, Microsatellite Instability (MSI) and Mismatch Repair Deficiency (dMMR)
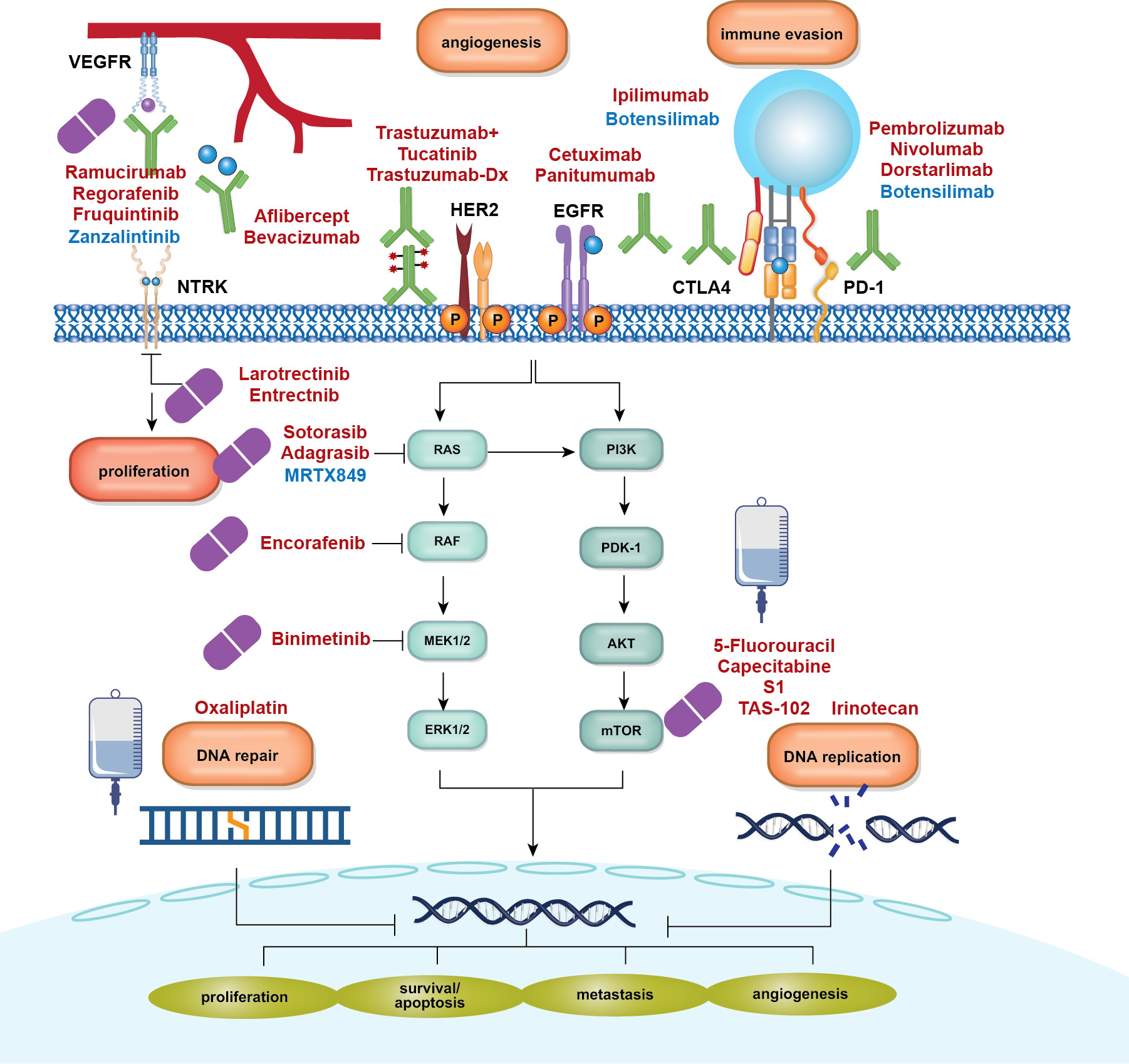
RAS targeted therapy in GI cancer
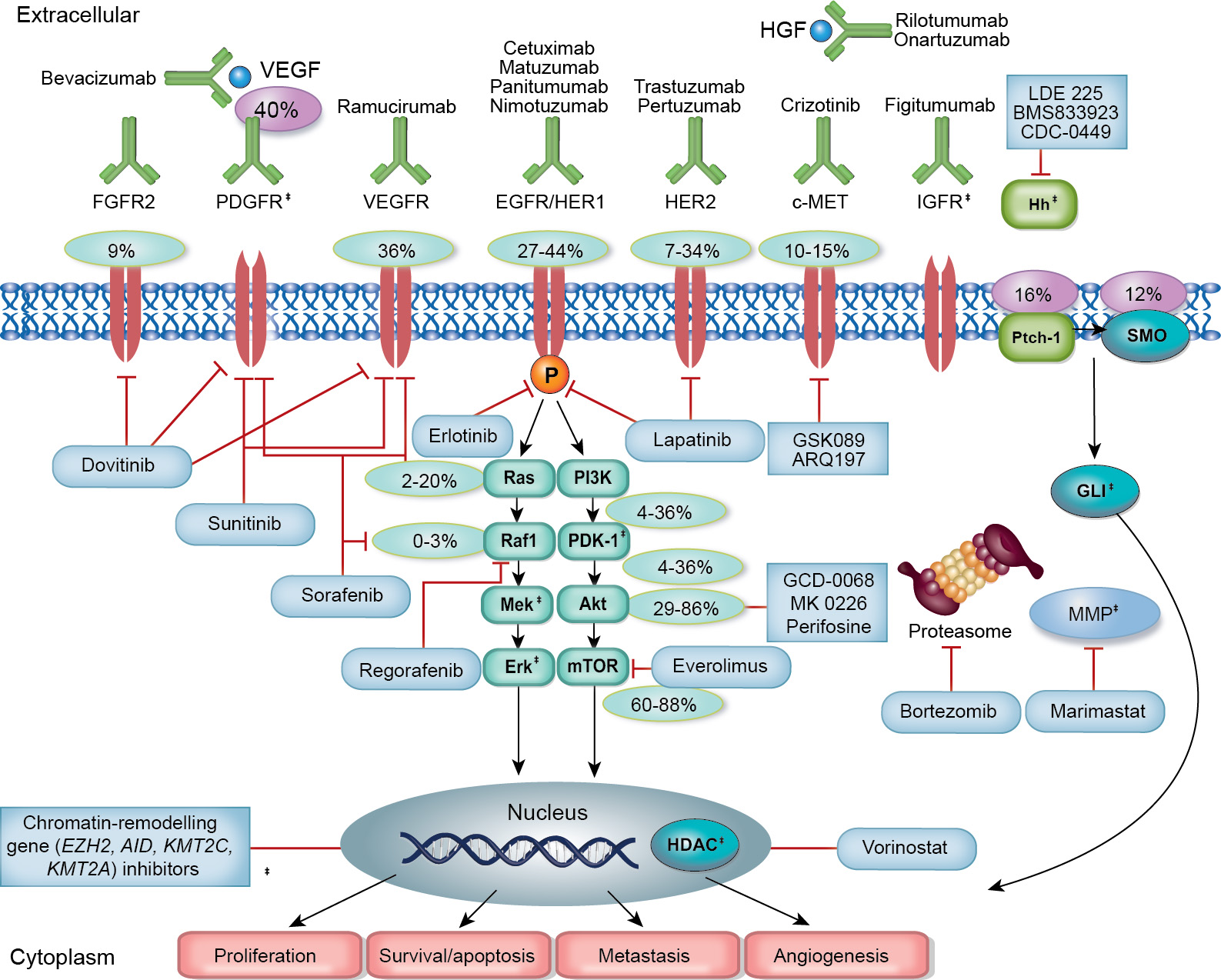
The RAS gene family (KRAS, NRAS, HRAS) plays a pivotal role in regulating cell growth, survival, and differentiation through the RAS–RAF–MEK–ERK (MAPK) signaling pathway. Mutations in this pathway, particularly in KRAS, result in constitutive activation, driving uncontrolled cell proliferation and resistance to standard therapies. KRAS mutations are among the most frequent oncogenic alterations in gastrointestinal cancers—occurring in ~40–50% of colorectal cancers (CRC), over 90% of pancreatic cancers, and a smaller subset of gastric cancers. NRAS mutations are rare in GI malignancies.
Historically considered “undruggable,” KRAS has become a viable target due to advances in covalent inhibitors that selectively bind the KRAS G12C mutant in its inactive GDP-bound state. Sotorasib (AMG 510) and adagrasib (MRTX 849) have demonstrated clinical efficacy, leading to recent FDA approvals—sotorasib plus panitumumab (2025) and adagrasib plus cetuximab (2024)—for KRAS G12C-mutated metastatic CRC.
Despite these breakthroughs, acquired resistance remains a major clinical challenge. Next-generation “RAS(ON)” inhibitors such as daraxonrasib and zoldonrasib are under active investigation, offering potential activity against a broader range of RAS-driven tumors. In parallel, drug development efforts are focusing on targeting other KRAS mutations, including G12D and G13D, which are highly prevalent in pancreatic and colorectal cancers.
These advances mark a transformative shift in GI oncology, enabling mutation-specific precision therapy and opening new therapeutic avenues for previously intractable cancers
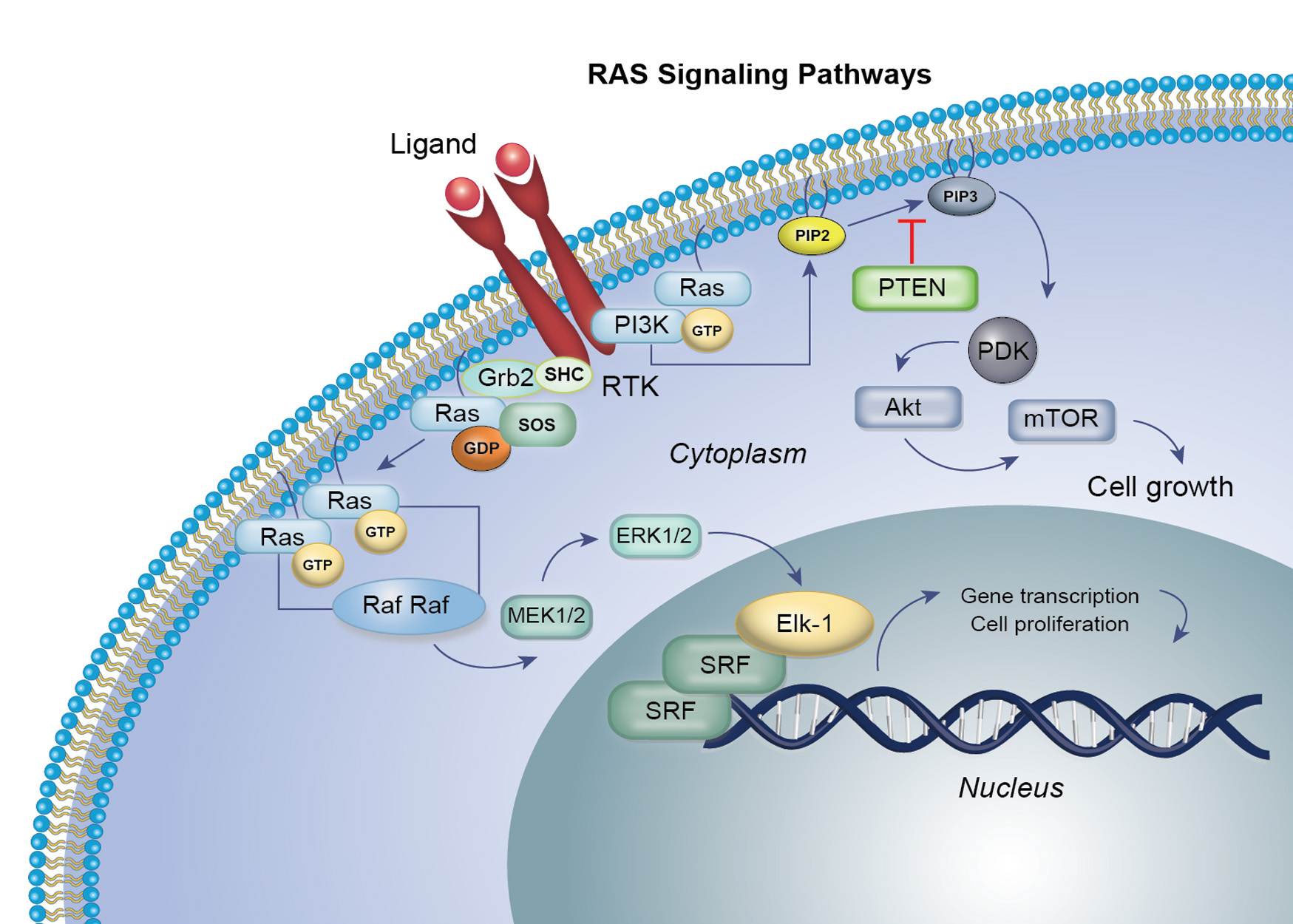
Exploring the role of different kinase inhibitors in GI cancer
Receptor tyrosine kinases (RTKs) such as KIT, PDGFRA, VEGFR, FGFR, EGFR, and HER2 are frequently dysregulated in gastrointestinal (GI) cancers, fueling tumor growth, angiogenesis, and metastatic spread.
Inhibitors of RAF kinases (e.g., vemurafenib, dabrafenib) , particularly those targeting the BRAF^V600E mutation, have shown significant clinical benefit in GI cancer, particularly CRC. resulting in improved rates of response and overall survival.
MEK1 and MEK2 are downstream kinases in the MAPK pathway and are critical effectors of BRAF and RAS signaling. The MEK inhibitors trametinib and cobimetinib have demonstrated clinical efficacy, particularly in combination with BRAF inhibitors. These combinations have been approved for use based on their ability to improve outcomes, including progression-free survival and overall survival, in these patients.
ERK1/2 are the terminal kinases in the MAPK cascade and have recently emerged as therapeutic targets with the development of ERK inhibitors. These inhibitors hold potential in overcoming resistance to upstream inhibitors and are currently being evaluated in clinical trials.
Beyond RTKs, mitotic kinases—such as Aurora kinases, polo-like kinase 1, and cyclin-dependent kinases play roles in GI tumor cell division and are under investigation as novel therapeutic targets.
On December 20, 2024, the Food and Drug Administration granted accelerated approval to encorafenib (Braftovi, Array BioPharma Inc., a subsidiary of Pfizer Inc.) with cetuximab and mFOLFOX6 for patients with metastatic colorectal cancer (mCRC) with a BRAF V600E mutation
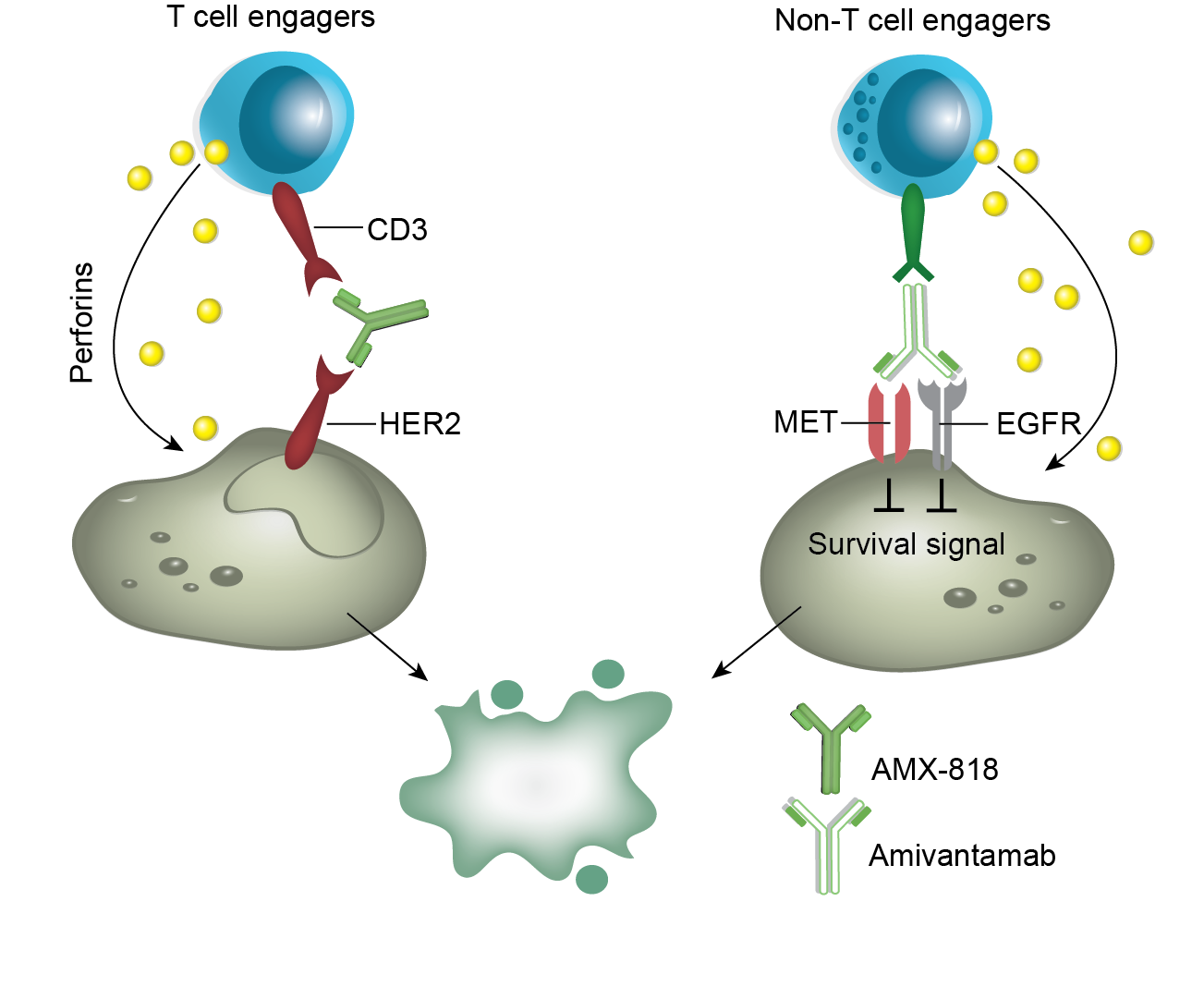
Targeting EGFR and MET in Metastatic Colorectal Cancer
The EGFR and MET receptor tyrosine kinases are central drivers of proliferation, survival, and invasion in metastatic colorectal cancer by activating downstream RAS/MAPK and PI3K/AKT pathways.
Targeted inhibition of EGFR—primarily using monoclonal antibodies such as cetuximab and panitumumab—is effective in patients with RAS wild-type tumors, improving survival and disease control
EGFR is upregulated in 60–80% of colorectal malignancies, and immunohistochemistry (IHC) assays often identify patients with EGFR expression in at least 1% of tumor cells. Activation of EGFR triggers intracellular signaling cascades, including MAPK and PI3K/Akt pathways, promoting proliferation, angiogenesis, and inhibition of apoptosis.
HER2 overexpression, seen in ~47.4% of CRC cases, is linked to poor prognosis, affecting differentiation, proliferation, and apoptosis, and contributing to chemoresistance through ERK1/2 signaling. HER2 also forms complexes with other ErbB family members, driving oncogenesis.
The proto-oncogene MET encodes c-MET, a receptor tyrosine kinase that, upon binding to hepatocyte growth factor (HGF), activates pathways like PI3K/AKT, RAS/RAF/ERK, STAT3, and NF-κB, fostering cellular growth and survival.
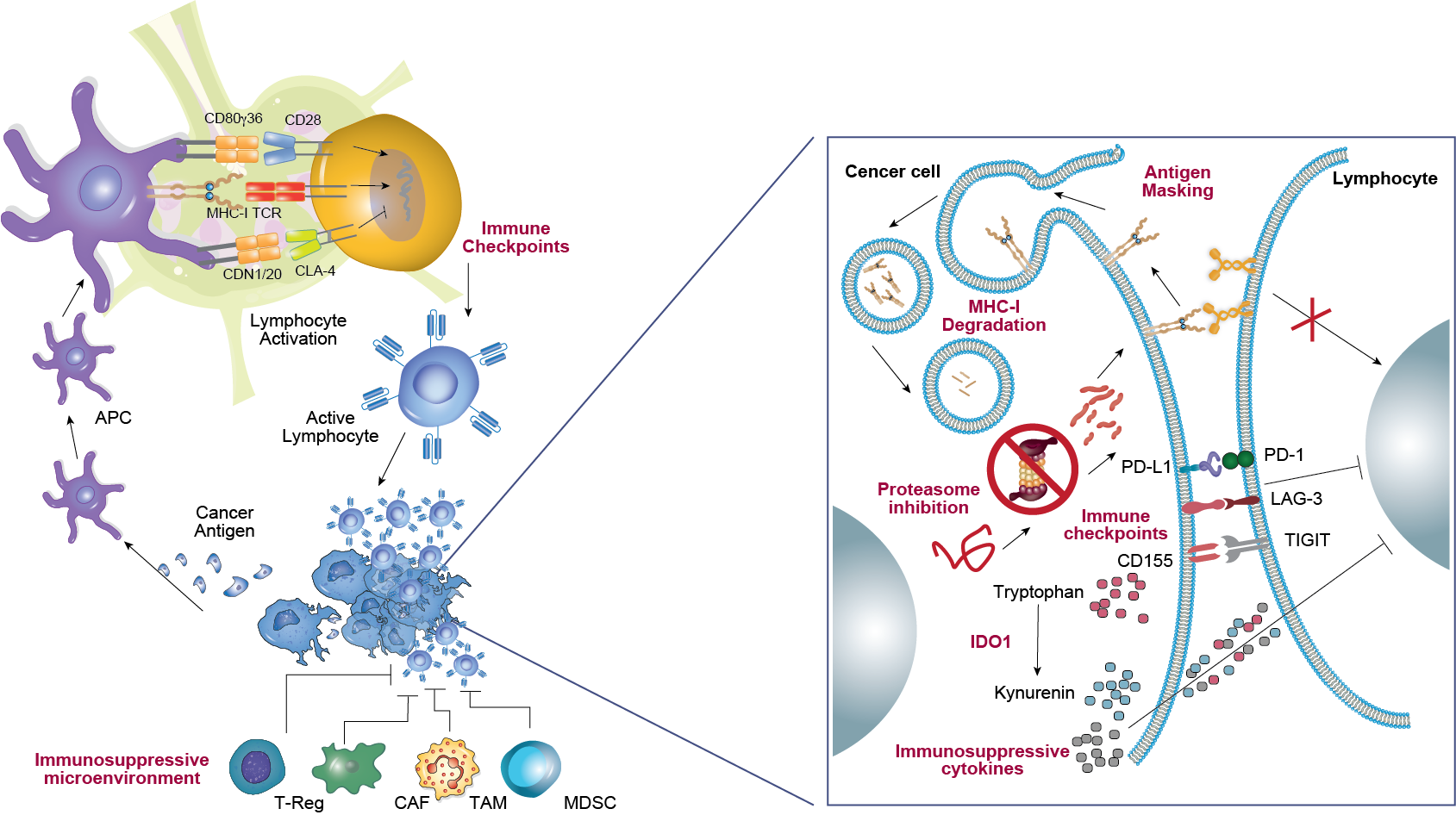
Immunotherapy in GI malignancies
Recent clinical trials of ICIs targeting PD-1 and PD-L1 have demonstrated significant efficacy in GI cancer, driving a paradigm shift in treatment approaches. Key trials, such as CheckMate 649, ORIENT-16, KEYNOTE 177, and KEYNOTE 590, have established anti-PD-1 antibodies combined with chemotherapy as first-line (1L) treatments for esophagogastric junction and gastric cancer, dMMR/MSI-H colorectal cancer, and advanced esophageal cancer.
The success of ICIs in first-line therapy has spurred numerous clinical trials exploring perioperative anti-PD-1/PD-L1 therapies, such as KEYNOTE 585 and MATTERHORN, to improve outcomes in resectable GI cancers. These trials aim to integrate immunotherapy earlier in the treatment continuum to enhance long-term benefits.
Beyond PD-1 and PD-L1, research is focusing on additional immune checkpoints, including TIGIT, LAG-3, CTLA-4, and CEACAM, to identify novel biomarkers and expand therapeutic targets in GI cancers. These advancements may help optimize patient selection and enhance treatment efficacy.
On December 27, 2024, the FDA approved tislelizumab in combination with chemotherapy for first-line treatment of gastric and gastroesophageal junction cancers, highlighting the ongoing evolution and clinical adoption of immunotherapy in GI oncology.
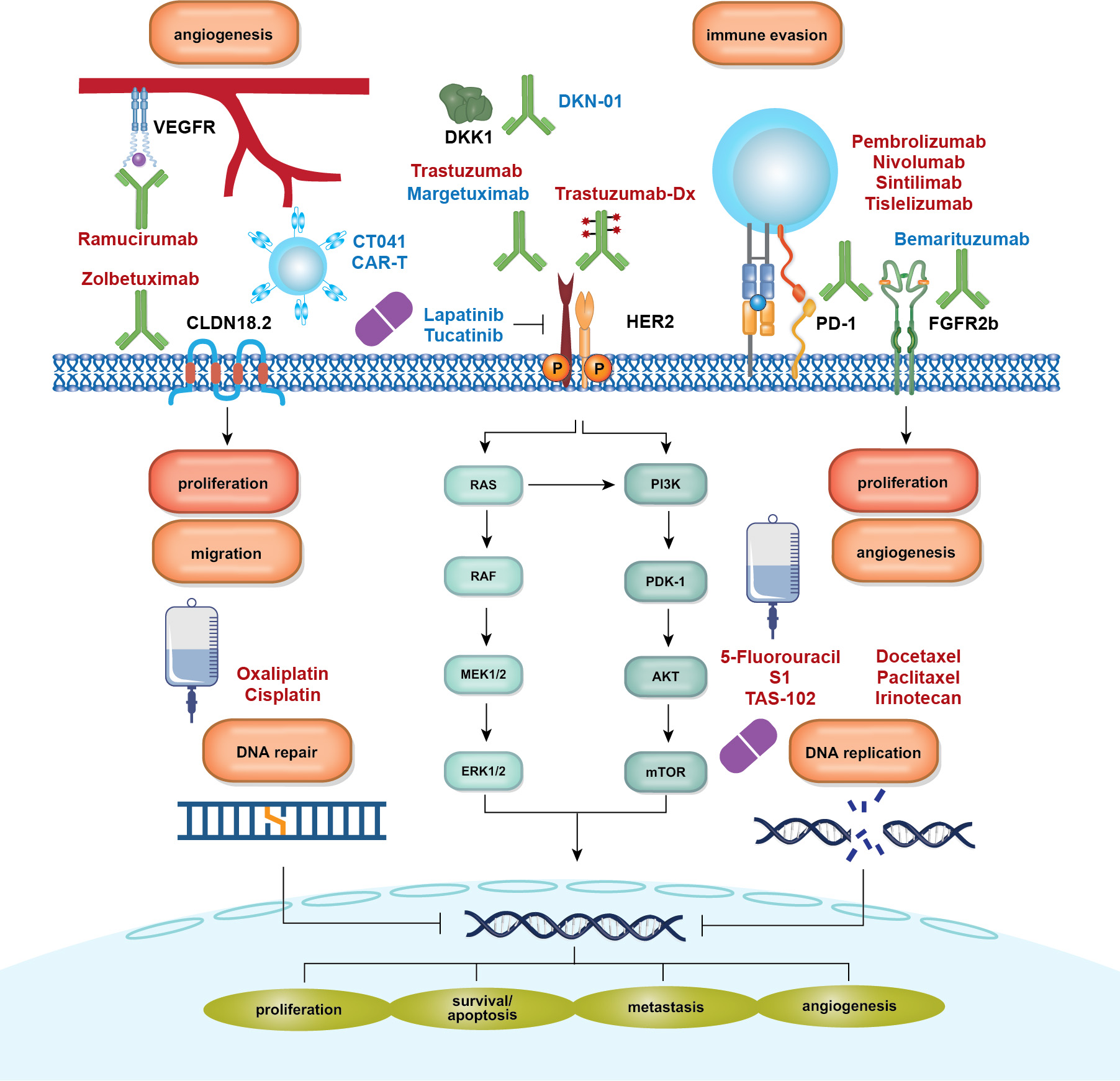
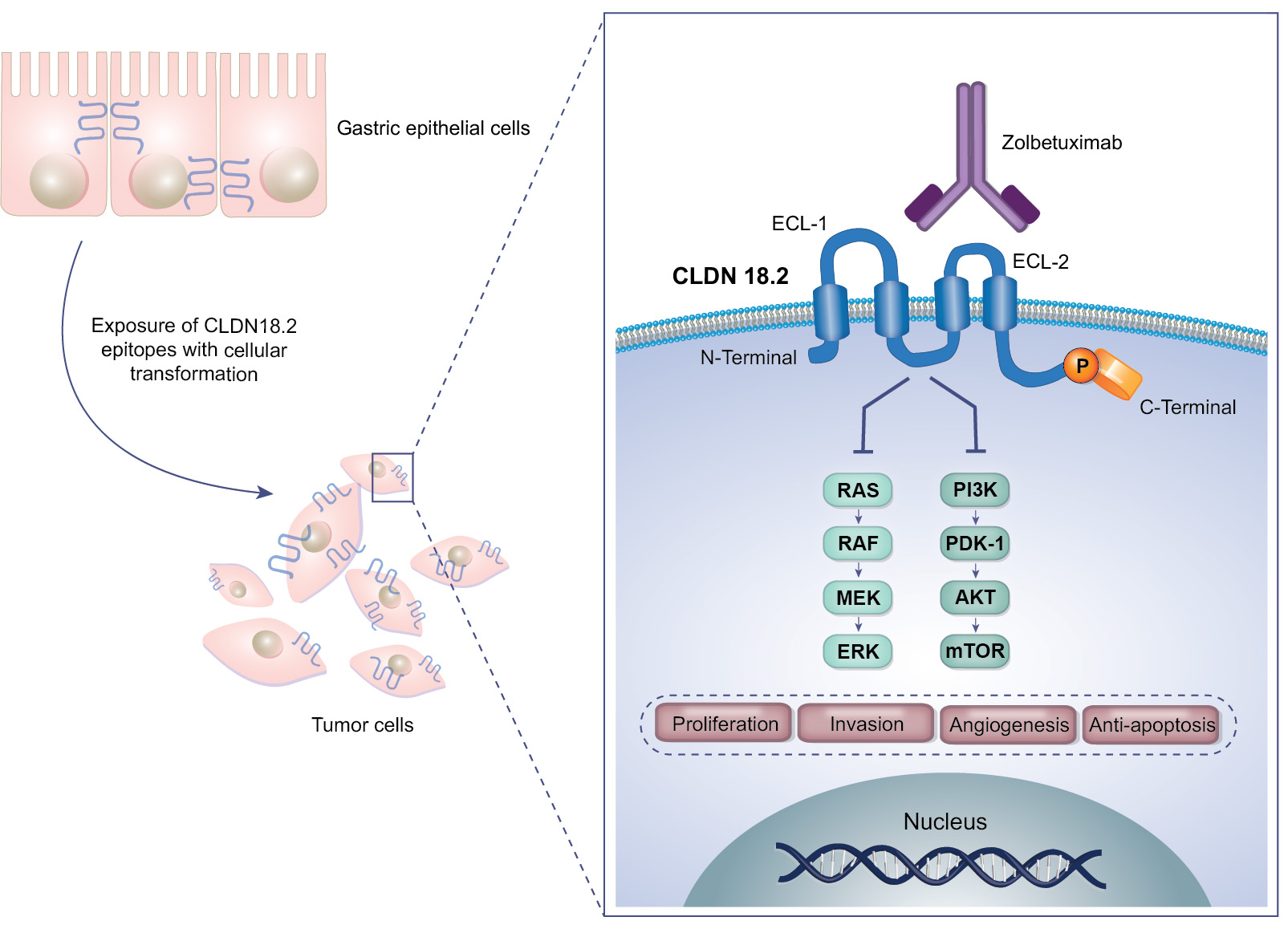
Targeting CLDN 18.2 in GC/GEJ Adenocarcinoma and other GI cancers
CLDN 18.2 is a tight-junction protein predominantly expressed in the gastric mucosa, with no definitive evidence linking it to the carcinogenesis or proliferation of gastric cancer. However, it has emerged as a promising therapeutic target due to its restricted expression and accessibility.
The monoclonal antibody zolbetuximab (Vyloy) targets CLDN18.2 and, in combination with chemotherapy, has been shown in the SPOTLIGHT and GLOW Phase III trials to improve both progression-free and overall survival in HER2‑negative, CLDN18.2‑positive GC/GEJ adenocarcinoma. The FDA approved zolbetuximab in October 2024.
CLDN 18.2 can be targeted with various therapeutic modalities, including bispecific antibodies, antibody–drug conjugates, CAR-T cells, and mRNA-based therapies. These innovative approaches are expanding the potential applications of CLDN 18.2-targeted treatments.
Additional Targeted Pathways and Agents in GI cancer
Angiogenesis, essential for tumor growth, is inhibited using VEGF-targeted agents like bevacizumab (monoclonal antibody) and tyrosine kinase inhibitors (e.g., sunitinib, sorafenib). These therapies benefit cancers like colorectal, lung, and renal cell carcinomas, forming a key component of anti-angiogenic strategies.
PARP inhibitors (e.g., olaparib, rucaparib, niraparib) target DNA repair deficiencies, especially in BRCA-mutated cancers. Initially approved for ovarian and breast cancers, their application is expanding across other cancer types with homologous recombination repair defects.
Novel therapeutic approaches include targeting the tumor microenvironment (TME), modulating the epigenome, and inhibiting proteasome or cancer metabolism. Promising agents targeting NTRK fusions (e.g., larotrectinib, entrectinib) and RET gene fusions (e.g., selpercatinib) have shown notable remission rates in specific colorectal cancer subsets.
The PI3K/AKT/mTOR pathway, crucial for cell growth, survival, and metabolism, is often dysregulated in cancer. Targeted inhibitors like PI3K inhibitors (e.g., idelalisib), AKT inhibitors (e.g., ipatasertib), and mTOR inhibitors (e.g., everolimus) have demonstrated efficacy in various cancers. Combination therapies are under evaluation to address the complexity and redundancy of this pathway and improve outcomes.
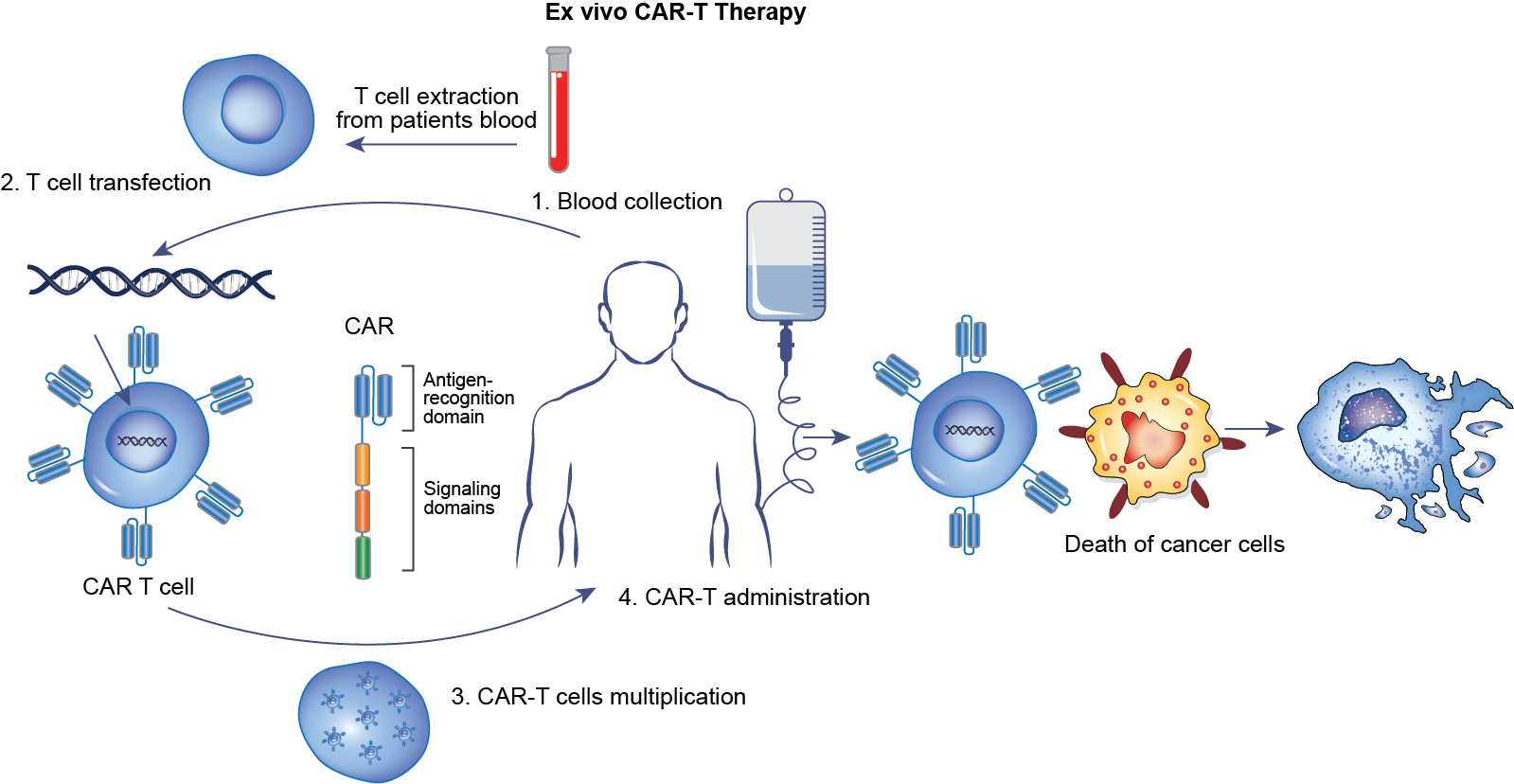
Cellular Therapies in Hepatocellular Carcinoma and other Solid tumors
CAR-T Cell Therapy is an innovative and promising treatment for HCC, leveraging genetic engineering to equip T cells with specific antibodies targeting cancer-associated antigens. This approach induces strong anti-tumor activity by directing T cells to attack malignant cells selectively.
Glypican-3 (GPC3), a protein overexpressed in HCC and other solid tumors, plays a pivotal role in cell proliferation, migration, and invasion, making it an ideal target for CAR-T therapy. Preclinical studies show that GPC3-targeting CAR-T cells effectively eliminate GPC3-positive HCC cells, induce tumor regression, and prolong survival in mouse models.
TCR therapy involves engineering T cells to express receptors specific for tumor-associated peptides presented by HLA molecules. This approach allows targeting of intracellular proteins, broadening the therapeutic horizon. Clinical trials are expanding for CAR-T and TCR therapies targeting novel antigens in gastric, colorectal, and hepatobiliary cancers. With advances in bioengineering, combination strategies with checkpoint inhibitors, and overcoming TME barriers, cellular therapies are poised to become a transformative option in GI cancer treatment.
Pexastimogene devacirepvec (Pexa‑Vec/JX‑594), an oncolytic vaccinia virus engineered to lyse tumor cells and stimulate immune responses via GM‑CSF expression, has demonstrated encouraging survival benefits (high‑dose vs. low‑dose) in phase II HCC trials, and is continuing in phase III when combined with sorafenib
ADCs and Bispecifics in Gastrointestinal
(GI) Cancer
Antibody–Drug Conjugates (ADCs) combine monoclonal antibodies (mAbs) with cytotoxic drugs to deliver anti-cancer agents specifically to malignant cells, minimizing systemic toxicity.
T-DXd has significantly reshaped the treatment of GI cancers, particularly gastric cancer, by demonstrating efficacy in advanced HER2-positive tumors. Its success in clinical trials led to FDA approval for HER2-positive gastric or gastroesophageal junction (GEJ) adenocarcinoma in patients previously treated with trastuzumab and chemotherapy.
Bispecific Antibodies target two distinct epitopes or antigens simultaneously, providing unique therapeutic advantages.
Zanidatamab (ZW25), a bispecific antibody that binds to two non-overlapping HER2 epitopes, has received FDA approval for first-line therapy in combination with chemotherapy for GEJ adenocarcinoma and as a single-agent treatment for refractory biliary tract cancer (BTC).